Polymers are inevitably exposed to sunlight or strong fluorescence during processing and use, resulting in a decline in their physical properties, yellowing, discoloration, embrittlement, and decreased transparency, which will not only affect the use experience, but also May cause serious consequences. Therefore, how to prevent polymer light aging has become an important research direction of polymer materials. At present, directly adding various light stabilizers to polymers is the most effective method to improve the light stability of materials. Common light stabilizers can be divided into light shielding agents, ultraviolet absorbers, quenchers and hindered amine light stabilizers according to their mechanism of action. Among them, hindered amine light stabilizers have the advantages of high efficiency, small addition amount, and less impact on products. , So it has won people’s favor. Hindered amine light stabilizers mainly act through four ways: decomposing hydroperoxide, quenching excited oxygen, capturing free radicals, and regenerating themselves, which can effectively improve the light stability of polymers.
Quick answer: A practical stabilization strategy starts with the degradation risk first, then screens the additive package around processing conditions, service life, and appearance requirements.
A very representative type of hindered amine light stabilizers is an organic amine compound with steric hindrance based on 2,2,6,6-tetramethyl-4-piperidinyl, 1972, Switzerland Ciba-Geigy (Ciba-Geigy) and Japan’s Sankyo company jointly developed and researched, and obtained two representative varieties of Light Stabilizer 744 and Light Stabilizer 770 through screening of 1,200 organic amine compounds. Light Stabilizer 770 has good compatibility with resin, no coloring, low toxicity, and good light stabilization effect. As an efficient light stabilizer, it is widely used in PP, PE, PVC, PS, ABS resin and other materials. At present, there are many reports on the application of light stabilizer 770, but the research on its synthesis process, especially the influence of the process on the product structure and components is less. This article will discuss the synthesis of light stabilizer 770, mainly involving the influence of different methods, solvents, catalyst types, post-treatment processes, etc. on the conversion rate and product performance.
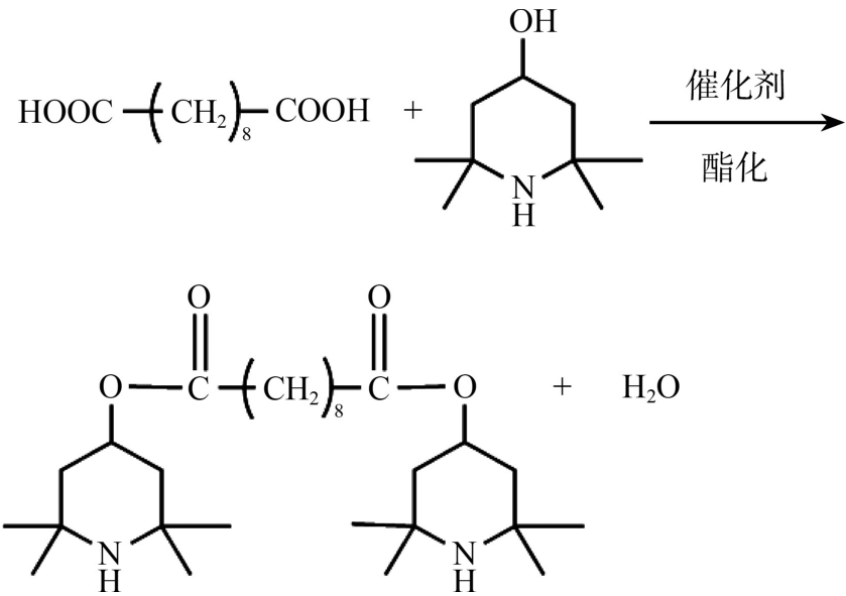
The generally used synthetic route is to use sebacic acid and tetramethylpiperidol to directly react dehydration and esterification to obtain the light stabilizer 770, as shown in the figure. According to whether a solvent is added, it can be divided into bulk method and solvent method.
1. In the bulk method, a certain proportion of sebacic acid, tetramethylpiperidinol and catalyst are added to the reaction vessel and heated, and the raw materials themselves will melt as a solvent for reaction. After the reaction, the product is washed with water or solvent and recrystallized to obtain the light stabilizer 770.
The choice of catalyst has a crucial influence on the reaction. A suitable catalyst can increase the activity of the reaction center and accelerate the reaction rate. General studies have shown that p-toluenesulfonic acid and phthalate esters have high catalytic efficiency for esterification, but when used in the synthesis of light stabilizer 770, the yields of both are less than 70%, mainly because of the system The raw material tetramethylpiperidinol is easy to sublime, which affects the conversion rate of raw materials. In addition, the reaction temperature should not be too high, which makes it difficult to remove the generated water, which will react with the phthalate ester catalyst to make it ineffective. Therefore, the researchers explored the solvent method to improve.
2. Solvent method is to add organic solvent on the basis of the above reaction. After the reaction, the solvent is evaporated and the product is processed. Compared with the bulk method, the solvent method does not need to reach the melting temperature of the reactants, reduces the reaction temperature of the system and reduces the sublimation phenomenon. At the same time, the water in the system can be taken out during the solvent reflux process and part of the sublimated raw materials can be brought back.
After exploring different solvents, it is found that petroleum ether and n-heptane below 100 °C have low efficiency. Although high boiling points of toluene and xylene can achieve high conversion rates, they are still unsatisfactory and have high boiling points. Solvent post-treatment is also more difficult.
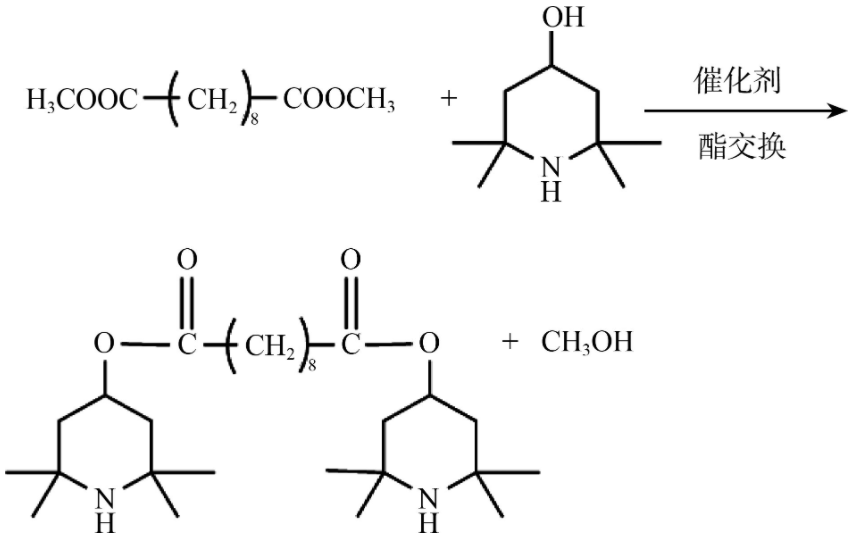
The researchers continued to explore the transesterification reaction route shown in the figure below. The reaction of dimethyl sebacate and tetramethylpiperidinol can be carried out at a lower temperature. The use of n-heptane as a solvent is beneficial to eliminate the reaction Methanol. Different catalysts have high catalytic efficiency, among which sodium catalysts have achieved a conversion rate of 99.8%. After further exploring the influence of the ratio of raw materials on the reaction, it was found that when the molar ratio of tetramethylpiperidinol to sebacic acid was 2.0:1, the conversion rate was the highest, and continuing to increase the ratio was not conducive to the later removal of excess raw materials.
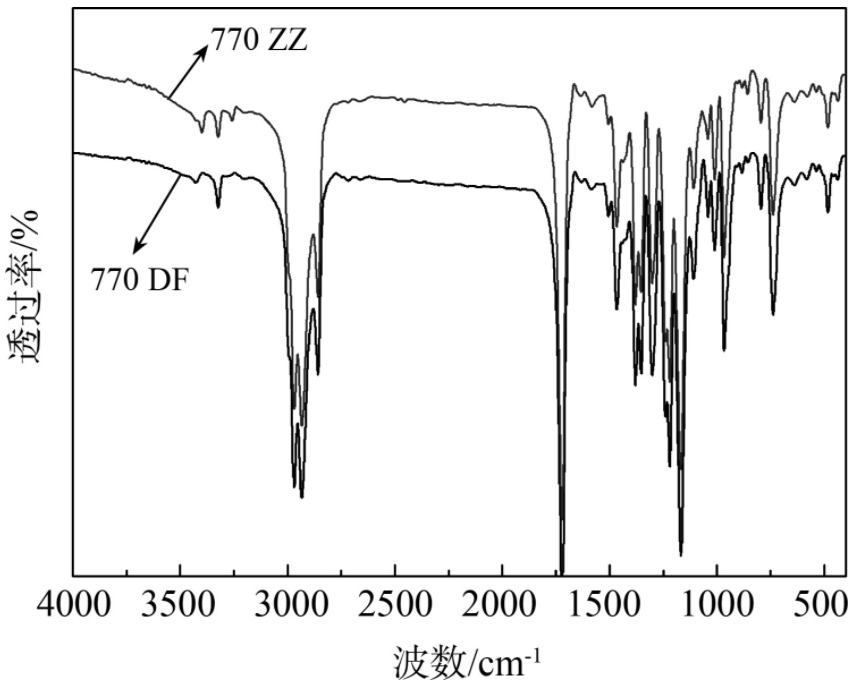
Finally, a self-made light stabilizer 770 (770 ZZ) was prepared based on the above exploration and compared with the commercial product (770 DF). The following figure is the infrared spectrum of the two. It can be seen that the characteristic absorption peaks of 770 DF and 770 ZZ are basically the same, and there are no other impurity peaks, indicating that the structure of the self-made light stabilizer 770 is consistent with the commercial product and has good purity.
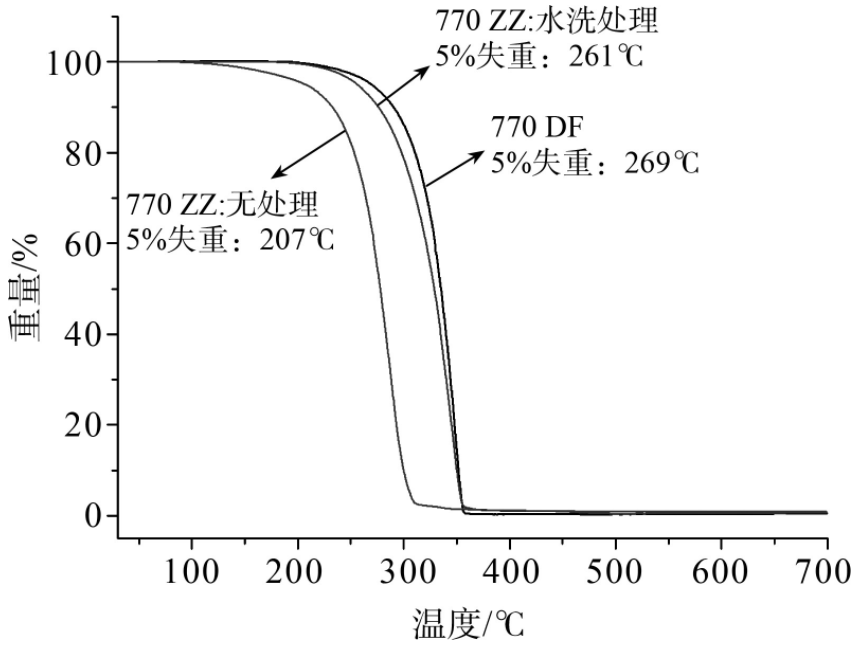
The figure below is the thermogravimetric curve. The light stabilizer 770, which has not been washed with water, has poor thermal stability. It may be that there is catalyst residue, which causes the product to undergo the reverse reaction of the esterification reaction at high temperature. The thermal stability of the processed product is not much different from that of commercial products, which can meet the processing requirements of most products.
Finally, the researchers tested the anti-ultraviolet performance of the self-made light stabilizer 770. After 672 hours of UV irradiation on ABS, the color of the swatch with 0.3 parts of 770 DF was slightly darker than the color of the swatch with 0.3 parts of 770 ZZ. Light stabilizer 770 has slightly better UV resistance. This may make the self-made products contain a small amount of monoesters that cannot be completely removed, which is beneficial to the dispersion of 770. In summary, the researcher’s self-made light stabilizer 770 has little difference in performance from commercial products. The influencing factors of the manufacturing process have been successfully explored and a route with higher yield has been obtained.
Contact Us Now!
We accept customized services, we will usually contact you within 24 hours. You could also email me info@longchangchemical.com during working hours ( 8:30 am to 6:00 pm UTC+8 Mon.~Sat. ) or use the website live chat to get prompt reply.
This article was written by Longchang Chemical R&D Department. If you need to copy and reprint, please indicate the source
A practical selection route for antioxidant, UV absorber, and HALS packages
Most stabilizer decisions work best when they are treated as package decisions rather than single-product decisions. Technical buyers usually get the strongest answer by reviewing long-term heat aging, process stability, weather exposure, and color sensitivity together.
- Separate processing protection from long-term stability: the best additive for melt history is not always the same one that gives the best service-life retention.
- Use synergy deliberately: many polymer and coating systems perform best when primary and secondary stabilizers are paired intentionally.
- Review color and clarity requirements: clear, pale, food-contact, or white systems often need a tighter package than dark industrial products.
- Check the real aging condition: heat, UV, humidity, and outdoor exposure can each change which stabilizer route is commercially strongest.
Recommended product references
- CHLUMILS UV-123: A strong HALS reference for weatherability-focused screens in coatings and polymers.
- CHLUMILS UV-5151: A practical stabilizer-package reference when broader light-aging protection is needed.
- CHLUMIFLEX ATBC: A practical non-phthalate plasticizer reference for application and compliance screens.
- CHLUMIFLEX DOTP: A standard terephthalate-plasticizer benchmark in flexible-plastics applications.
FAQ for buyers and formulators
Why are stabilizer packages often stronger than a single additive?
Because different products can protect different parts of the degradation pathway, so the package often covers more risk than one grade alone.
Does adding more antioxidant or UV stabilizer always improve performance?
Not necessarily. Over-dosing can increase cost and sometimes create side effects, so most systems perform best inside a tested dosage window.

Ann-Marie Garrard Besse
Some truly good articles on this internet site , appreciate it for contribution.
Aimil Farlie Frazer
I am genuinely thankful to the holder of this website who has shared this wonderful article at here.
Siobhan Calhoun Ana
Generally I do not learn article on blogs, however I would like to say that this write-up very pressured me to take a look at and do so! Your writing taste has been amazed me. Thank you, quite great post.